The degree to which the central nervous system function is impaired is directly proportional to the concentration of alcohol in the blood. Alcohol is an organic compound composed of carbon, oxygen, and hydrogen its chemical formula is C2H5OH.Īlcohol is a central nervous system depressant and it is the central nervous system which is the bodily system that is most severely affected by alcohol (see chart below). It has a slight, characteristic odor and is very soluble in water. This introduction discusses the physical, chemical, and physiological aspects of the most commonly ingested of these - ethanol.Īlcohol (ethanol) is a clear, volatile liquid that burns (oxidizes) easily. Members of this family include ethanol, methanol, isopropanol, and others. Alcohol's PropertiesĪlcohol is a general term denoting a family of organic chemicals with common properties. Therefore, as far as their alcoholic contents are concerned, a can of beer, a glass of wine and a shot of whiskey are all the same. At a typical concentration of 40 percent ethanol (80-proof), the standard shot of whiskey has approximately one-half ounce of ethanol. Whiskey and other distilled spirits are dispensed by the "shot glass", usually containing about one and one-quarter ounce of fluid. Wine is about 12 percent alcohol, so the glass of wine also has a bit less than one-half ounce of ethanol in it. A standard glass of wine has about four ounces of liquid. Since beer has an ethanol concentration of about four percent, the typical bottle or can of beer contains a little less than one-half ounce of pure ethanol. Beer, for example, is normally dispensed in 12-ounce servings. Over the millennia during which people have used and abused ethanol, some standard-size servings of the different beverages have evolved. For example, an 80-proof beverage has an ethanol concentration of 40 percent. The ethanol concentration of distilled spirits usually is expressed in terms of proof, which is a number corresponding to twice the ethanol percentage. These include rum, whiskey, gin, vodka, etc. It is possible to do this because ethanol boils at a lower temperature than does water.ĭistilled spirits is the name we give to high-ethanol-concentration beverages produced by distillation. This involves heating the beverage until the ethanol "boils off", then collecting the ethanol vapor. Obtaining a higher ethanol content requires a process called distillation. At that concentration, the yeast dies, so the fermentation stops. When an alcoholic beverage is produced by fermentation, the maximum ethanol content that can be reached is about 14 percent. However, most of the ethanol in the world didn't ferment naturally, but was produced under human supervision. This can occur naturally, as yeast spores in the air come into contact with decomposing fruit and grains. That is a kind of decomposition in which the sugars in fruit, grains and other organic materials combine with yeast to product the chemical we call ethanol. Ethanol production starts with fermentation. Ethanol is beverage alcohol, the active ingredient in beer, wine, whiskey, liquors, etc. Two of ethanol's best known analogs are methyl alcohol (or methanol), commonly called "wood alcohol", and isopropyl alcohol (or isopropanol), also known as "rubbing alcohol".Įthanol is what interests us, because it is the kind of alcohol that features prominently in impaired driving. Ethanol is the variety of alcohol that has two carbon atoms. The "ET" stands for "ethyl" and the "OH" represents the single oxygen atom and one of the hydrogen atoms, bonded together in what chemists refer to as the "hydroxy radical". The ingestible alcohol is known as ethyl alcohol, or ethanol. 
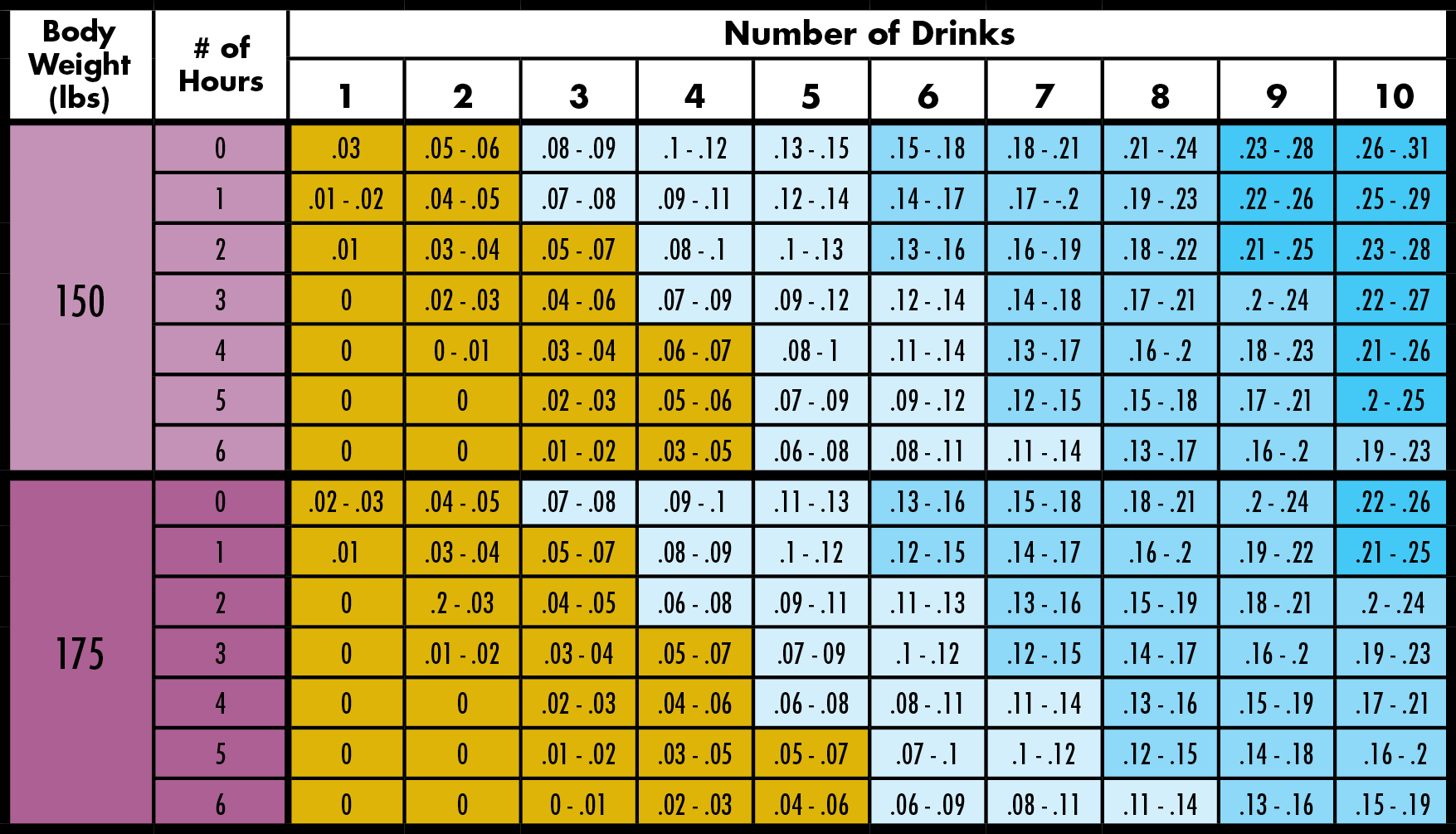
This is done directly when dealing with a blood sample, or indirectly by analyzing the percentage in a breath sample and applying conversion ratios to estimate the percentage in the blood these conversion ratios used in DUI cases are inaccurate in that they simply represent statistical averages. Careless Driving Causing Bodily Injury or DeathīAC Physiology Blood & Breath Alcohol Physiologyīlood-alcohol analysis is simply the attempt to measure the percentage by weight of alcohol within the DUI suspect's blood at the time of testing.How to Protect Your Rights During a DUI Stop.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
